PharmSmart: Phenytoin Phacts (Dilantin)
By AccentCare
GG is a 67 year old female who was admitted to hospice care 3 weeks ago with a diagnosis of breast cancer with metastasis to the brain. The patient has had several seizures, thought to be related to the brain mets, but controlled on phenytoin. She weighs 110 pounds (50 kg) and is 5’6” tall. Past medical history is positive for hypertension, dyslipidemia, COPD, cirrhosis, deep vein thrombosis (5 years ago), eczema, and depression. She has smoked 3 packs of cigarettes per day for the past 35 years.
Her current medications include:
- Hydrochlorothiazide 25 mg po once daily
- Lisinopril 20 mg po once daily
- Betamethasone valerate topical cream 0.1% applied once or twice daily
- Albuterol/ipratropium (Combivent), 2 inhalations by mouth four times daily
- Bupropion 300 mg po once daily
- Simvastatin 20 mg po once daily
- Phenytoin sodium 300 mg XL (Dilantin) capsule once daily
When you make a visit on a Tuesday morning (you saw the patient last Thursday) her husband tells you the patient has been experiencing many symptoms the past few days: dizziness, severe nausea and vomiting, an inability to sleep well (due to vivid hallucinations) and a headache that just won’t quit. Also, she appears to be walking more slowly than usual, with an unsteady gait. Her physician asks that you draw a phenytoin serum level, which comes back as 15 mcg/ml (therapeutic is 10-20 mcg/ml).
Which of the following do you is MOST LIKELY responsible for GG’s symptoms?
- Orthostatic hypotension
- Phenytoin toxicity
- Bupropion toxicity
- Metastatic breast cancer
If you picked “B” you are clearly smarter than the average bear!
Phenytoin is primarily absorbed from the duodenum and it passes quickly into the brain before being redistributed to other body tissues. Phenytoin is a highly bound (90%) to the serum protein albumin. When drawing a serum phenytoin concentration, it is important to realize that you should request a TOTAL phenytoin concentration (bound and unbound to albumin), and an “unbound” or “free” phenytoin concentration in addition to a total phenytoin concentration.
The most IMPORTANT measurement is the “free fraction” (or “unbound”) phenytoin serum concentration (therapeutic is 1 – 2 mcg/ml; this represents the 10% unbound of the total therapeutic concentration of 10-20 mcg/ml). Only the “unbound” or “free” phenytoin portion is able to interact with the target tissues to exert therapeutic OR toxic effects; this is USUALLY 10% of the total serum phenytoin concentration. However when serum albumin is decreased (hypoalbuminemia) less phenytoin is bound to albumin, and more drug is FREE or UNBOUND. Obviously, this increases the risk for phenytoin toxicity, while the TOTAL serum phenytoin concentration remains the same.
Our patient GG has a TOTAL serum phenytoin concentration of 15 mcg/ml. With adequate albumin stores, 90% of the phenytoin in the body is bound to serum albumin; the remaining 10% is “unbound” or “free.” If GG had a normal albumin level, we could expect her “free” phenytoin level to be 1.5 mcg/ml. Let’s take a look at this graphically.
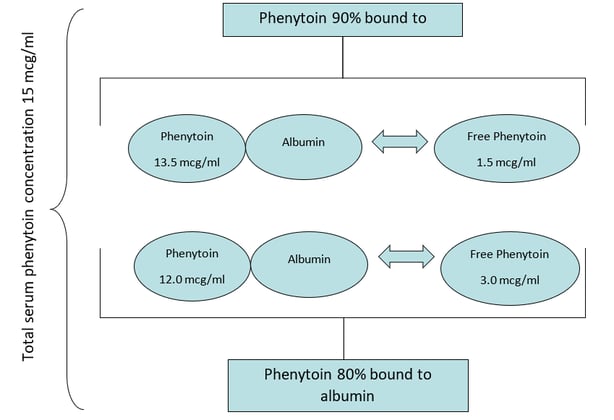
As shown in the picture above, if GG had a reduced albumin level (hypoalbuminemia), MORE of the phenytoin in the body is “unbound” or “free” (not bound to albumin). Again, this is the phenytoin in the body that controls seizures and may cause side effects. If 20% instead of 10% of the total phenytoin is “unbound” this DOUBLES the amount of pharmacologically active phenytoin in the blood. Therefore, despite a “therapeutic” total phenytoin level of 15 mcg/ml, if the patient has a low albumin more phenytoin is unbound, and this puts the patient at risk for toxicity. It is not unlikely that patients with advanced illness (e.g., hospice patients) have hypoalbuminemia due to liver dysfunction/disease, malnutrition, cachexia or even heart failure.
A “free” or “unbound” phenytoin serum level can be calculated based on the patient’s serum albumin levels: [patient’s phenytoin concentration]/[(0.9)*(patient’s serum albumin/4.4 g/dl)+0.1)]. However, it is preferable to have a “total” and “unbound” or “free” phenytoin concentration assayed by the lab. For example, if GG’s physician had asked you to order a total and free phenytoin serum concentration, you may have gotten the following results:
- Total phenytoin 15 mcg/ml (therapeutic 10 – 20 mcg/ml)
- Free phenytoin 3 mcg/ml (therapeutic 1 – 2 mcg/ml)
- Albumin 2.2 g/dl (normal 3.5 – 5 g/dl)
In this case, you would reduce GG’s phenytoin dose, and recheck the “unbound” serum level is about a week (once steady-state is achieved) and hope it is in the therapeutic range of 1-2 mcg/ml.
Phenytoin has “saturable pharmacokinetics,” meaning the liver enzymes responsible for the metabolism of phenytoin become saturated quickly. Once saturation is achieved no further phenytoin metabolism can occur until more enzyme sites become available for binding to the drug. Therefore the remaining serum phenytoin is free to circulate and produce effects within the body. This liver enzyme saturation can occur at any serum phenytoin concentration and may occur at therapeutic doses. Any dosage changes, even slight modifications, may result in unpredictable serum concentrations leading to exaggerated effects of toxicity or inadequate disease control.
Speaking of changing dosage formulations, phenytoin is available as a parenteral formulation and several oral formulations. Dilantin extended release capsules and Dilantin parenteral is formulated as the sodium salt of phenytoin. If a patient has difficulty swallowing and you want to switch to the Dilantin-125 suspension or Dilantin Infatabs, it’s important to know this is the free acid form of phenytoin. This results in about an 8% increase in drug content with the free acid form (suspension and Infatabs) vs. the sodium salt (extended release capsule and parenteral). This doesn’t sound like a big difference but sometimes that 8% can result in a supratherapeutic phenytoin level and toxicity! ALSO, the only Dilantin formulation that can be given once daily is the extended release Kapseal capsule. The oral suspension and Infatabs must be divided into three doses and administered every 8 hours.
A few last points to remember about phenytoin include the following:
- Always instruct patients to shake phenytoin suspension very well prior to use to assure equal distribution of the drug.
- Only Dilantin Kapseals brand can be administered once daily. ALL other formulations of phenytoin must be administered three times daily. This includes generic capsules, chewable tablets, and liquid suspension.
- When administering oral phenytoin suspension concurrently with continuous nasogastric feedings, phenytoin absorption is reduced. To minimize this, hold the continuous enteral feeding for 1-2 hours before and after phenytoin administration.
Self-Assessment Questions
- What is the therapeutic range of phenytoin?
- Total of 5-15 mcg/ml; free of 1-2 mcg/ml
- Total of 5-20 mcg/ml; free of .5-1.5 mcg/ml
- Total of 10-20 mcg/ml; free of 1-2 mcg/ml
- Total of 15-25 mcg/ml; free of 1.5-3 mcg/ml
- What percentage of phenytoin in the body is bound to albumin (assuming normal albumin)?
- 10%
- 25%
- 50%
- 90%
- 100%
- If a patient were receiving Dilantin Kapseals 300 mg by mouth once daily, and needs to switch to the Dilantin-125 oral suspension, what dose would you recommend?
- 90 mg po q8h
- 100 mg po q8h
- 270 mg po q24h
- 300 mg po q24h
Answers:
1. C; 2. D; 3. A
PharmSmart is a monthly article dedicated to best practices in drug management for patients nearing the end of life, with a little cheer and lightheartedness woven throughout. It is edited by Dr. Mary Lynn McPherson, PharmD. Dr. McPherson is the Executive Director of Advanced Post-Graduate Education in Palliative Care at University of Maryland. Dr. McPherson is a consultant pharmacist to Seasons, and answers complex medication questions for our clinical teams at all hours of the day or night. She is a nationally-recognized expert in medication management for hospice and palliative care patients.